Composition and Layers of the Atmosphere
It's our atmosphere that makes life on Earth possible. Not only does the air provide the oxygen we need to breath, but it also serves other important functions.
One of those is that the atmosphere moderates our planet's temperature, reducing the extremes that occur on airless worlds. For example, temperatures on the moon range from 120 °C, or about 250 °F, (see the online temperature converter) in the day to -170 °C (about -275 °F) at night.
The atmosphere also protects us by absorbing and scattering harmful radiation from the sun and space.
Table of Contents
WHAT IS THE ATMOSPHERE?
The atmosphere is the mixture of gases and other materials that surround the Earth in a thin, mostly transparent shell. It is held in place by the Earth's gravity. The main components are nitrogen (78.09%), oxygen (20.95%), argon (0.93%), and carbon dioxide (0.03%). The atmosphere also contains small amounts, or traces, of water (in local concentrations ranging from 0% to 4%), solid particles, neon, helium, methane, krypton, hydrogen, xenon and ozone.
The study of the atmosphere, particularly weather and climate, is called meteorology.
Of the total amount of the sun's energy that reaches the Earth, 30% is reflected back into space by clouds and the Earth's surface. The atmosphere absorbs 19%. Only 51% is absorbed by the Earth's surface.
We are not normally aware of it but air does have weight. The column of air above us exerts pressure on us. This pressure at sea level is defined as one atmosphere. Other equivalent measurements you may hear used are 1,013 millibars, 760 mm Hg (mercury), 29.92 inches of Hg, or 14.7 pounds/square inch (psi). (See the online converter for pressure.) Atmospheric pressure decreases rapidly with height. Pressure drops by a factor of 10 for every 16 km (10 miles) increase in altitude. This means that the pressure is 1 atmosphere at sea level, but 0.1 atmosphere at 16 km and only 0.01 atmosphere at 32 km.
The density of the lower atmosphere is about 1 kg/cubic meter (1 oz./cubic foot). There are approximately 300 billion billion (3 x 10**20, or a 3 followed by 20 zeros) molecules per cubic inch (16.4 cubic centimeters). At ground level, each molecule is moving at about 1600 km/hr (1000 miles/hr), and collides with other molecules 5 billion times per second.
The density of air also decreases rapidly with altitude. At 3 km (2 miles) air density has decreased by 30%. People who normally live closer to sea level experience temporary breathing difficulties when traveling to these altitudes. The highest permanent human settlements are at about 4 km (3 miles).
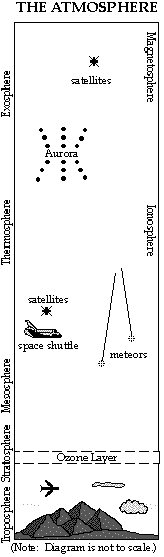
LAYERS OF THE ATMOSPHERE
The atmosphere is divided into layers based on temperature, composition and electrical properties. These layers are approximate and the boundaries vary, depending on the seasons and latitude. (The boundaries also depend on which "authority" is defining them.)
LAYERS BASED ON COMPOSITION
Homosphere
- The lowest 100 km (60 miles), including the Troposphere, Stratosphere and Mesosphere.
- Contains 99% of the atmosphere's mass.
- Molecules do not stratify by molecular weight.
- Although small local variations exist, it has a relatively uniform composition, due to continuous mixing, turbulence and eddy diffusion.
- Water is one of two components that is not equally distributed. As water vapor rises, it cools and condenses, returning to earth as rain and snow. The Stratosphere is extremely dry.
- Ozone is another molecule not equally distributed. (Read about the ozone layer in the Stratosphere section below.)
Heterosphere
- Extends above homosphere, including the Thermosphere and Exosphere.
- Stratified (components are separated in layers) based on molecular weight. The heavier molecules, like nitrogen and oxygen, are concentrated in the lowest levels. The lighter ones, helium and hydrogen, predominate higher up.
LAYERS BASED ON ELECTRICAL PROPERTIES
Neutral atmosphere
- Below about 100 km, or about 60 miles. (See the online distance converter)
Ionosphere
- Above about 100 km.
- Contains electrically charged particles or ions, created by the absorption of UV (ultraviolet) light.
- The degree of ionization varies with altitude.
- Different layers reflect long and short radio waves. This allows radio signals to be sent around the curved surface of the earth.
- The Aurora Borealis and Aurora Australis (the Northern and Southern Lights) occur in this layer.
- The Magnetosphere is the upper part of the ionosphere, extending out to 64,000 km (40,000 miles.) It protects us from the high energy, electrically charged particles of the solar wind, which are trapped by the Earth's magnetic field.
LAYERS BASED ON TEMPERATURE
Troposphere
- Height depends on the seasons and latitude. It extends from ground level up to about 16 km (10 miles) at the equator, and to 9 km (5 miles) at the North and South Poles.
- The prefix "tropo" means change. Changing conditions in the Troposphere result in our weather.
- Temperature decreases with increasing altitude. Warm air rises, then cools and falls back to Earth. This process is called convection, and results in huge movements of air. Winds in this layer are mostly vertical.
- Contains more air molecules than all the other layers combined.
Stratosphere
- Extends out to about 50 km (30 miles).
- The air is very thin.
- The prefix "strato" is related to layers, or stratification.
- The bottom of this layer is calm. Jet planes often fly in the lower Stratosphere to avoid bad weather in the Troposphere.
- The upper part of the Stratosphere holds the high winds known as the jet streams. These blow horizontally at speeds up to 480 km/hour, or about 300 miles/hour.
- Contains the "ozone layer" located between 15 - 40 km ( 10 - 25 miles) above the surface. Although the concentration of ozone is at most 12 parts per million (ppm), it is very effective at absorbing the harmful ultraviolet (UV) rays of the sun and protecting life on Earth. Ozone is a molecule made of three oxygen atoms. The oxygen molecule we need to breathe contains two oxygen atoms.
- The temperature is cold, about -55 °C (-67 °F) in the lower part, and increases with increasing altitude. The increase is caused by the absorption of UV radiation by the oxygen and ozone.
- The temperature increase with altitude results in a layering effect. It creates a global "inversion layer", and reduces vertical convection.
Mesosphere
- Extends out to about 100 km (65 miles).
- Temperature decreases rapidly with increasing altitude.
Thermosphere
- Extends out to about 400 km ( 250 miles).
- Temperature increases rapidly with increasing altitude, due to absorption of extremely short wavelength UV radiation.
- Meteors, or "shooting stars," start to burn up around 110-130 km (70-80 miles) above the earth.
Exosphere
- Extends beyond the Thermosphere hundreds of kilometers, gradually fading into interstellar space.
- Density of the air is so low that the normal concept of temperature loses its meaning.
- Molecules often escape into space after colliding with one another.
